Viruses are found wherever there is life and have probably existed since living cells first evolved. Most virus infections eventually result in the death of the host cell. Viral infections provoke an immune response that usually eliminates the infecting virus. However, some viruses including those causing AIDS, viral hepatitis, genital warts and cervical cancer evade these immune responses and result in chronic infections. Antibiotics have no effect on viruses. Viral populations do not grow through cell division, because they are acellular. Instead, they use the machinery and metabolism of a host cell to produce multiple copies of themselves, and they assemble in the cell.
Some viruses cause no apparent changes to the infected cell. Cells in which the virus is latent and inactive show few signs of infection and often function normally. This causes persistent infections and the virus is often dormant for many months or years. This is often the case with herpes viruses. Some viruses, such as Epstein-Barr virus, can cause cells to proliferate without causing malignancy, while others, such as papillomaviruses (HPVs), hepatitis B and C viruses are established causes of cancer.
Unlike most bacteria, which can be recognized by their distinctly foreign features, a virus becomes invisible as it hides and multiplies inside the infected cells of the host. To enable the immune system to detect viruses, the host cells have evolved an elaborate mechanism called the “antigen presentation pathway. This pathway scrutinizes virtually all actively synthesized proteins in the cell. A few snippets or peptides are produced from the proteins and are ferried to the cell surface by the major histocompatibility complex (MHC) class I molecules. The circulating T cells of the immune system probe the peptide–MHC complexes (pMHCs) on the cell surface to identify and eliminate infected cells. Viruses have evolved various strategies to prevent peptide display by MHC molecules. If any of the key steps in the antigen presentation pathway is blocked, MHC molecules fail to present appropriate peptides.
The intracellular antigen transport machinery TAP in adaptive immunity and virus escape mechanisms.
Immune surveillance obstructed by viral mRNA.
The MHC class I antigen presentation pathway: strategies for viral immune evasion.
MHC class I antigen presentation: learning from viral evasion strategies.
HPV infections are exclusively intra-epithelial and, theoretically, HPV attack should be detected. Regression of HPV infections is accompanied histologically by a CD4+ T cell dominated Th1 response. However, HPV avoids host defences. How HPV infection remains undetected by the immune system for so long? In this location there is only a limited expression of viral proteins. Other factors contributing to the low level of host immunity are that HPV infection is non-lytic (does not cause death of the infected cell); that a functionally active immune response is generated only at later stages of HPV infection, in post-mitotic suprabasal keratinocytes where all viral genes are expressed; and that only in suprabasal keratinocytes has the HPV DNA been sufficiently amplified to be detected by the host immune-surveillance cells.
The prolonged duration of HPV infections is associated with effective evasion of innate immunity as reflected in the absence of inflammation during virus replication, assembly and release and down regulation of interferon secretion and response thus delaying the activation of adaptive immunity. Thus,for most of the duration of the HPV infectious cycle there is little or no release into the local milieu of proinflammatory cytokines important for DC (dendritic cells) activation and migration and the central signals to kick start the immune response in squamous epithelia are absent.
In addition to the natural low-level immune responses towards HPV, HPV also employs several mechanisms to down-regulate innate and cell-mediated immunity, thus facilitating host immune evasion and persistent infection. Tregs (regulatory T cells: known as suppressor T cells) are a specialized subpopulation of T cells that act to suppress activation of the immune system and thereby maintain immune system homeostasis and tolerance to self-antigens. It is believed that Tregs suppress the immune control of cervical neoplasia and furthermore that suppression of immunity by Tregs will be another hurdle to overcome in therapeutic immunization strategies against cervical neoplasia. The environment, lifestyle, the genetic make-up of the host, and the viral genomic characteristics can also influence the persistence of HPV infection, and consequential diseases.
Immune responses to human papilloma viruses.
Frequencies and role of regulatory T cells in patients with (pre)malignant cervical neoplasia.
HPV infections disrupt cytokine expression and signalling with the E6 and E7 oncoproteins particularly targeting the type I interferon (IFN) pathway. High doses of IFN can overcome the HPV-mediated abrogation of signalling, and this may be the basis for the therapeutic effects on HPV infections of immune-response modulators. Stimulation of TLRs (Toll-like receptors) results in the activation of innate immune cells such as macrophages and dendritic cells, leading to the production of proinflammatory cytokines and up-regulation of costimulatory molecules. Thus, TLRs provide the “danger signals” required for clearance of infection by innate immune cells and also for the initiation of adaptive immune responses. Activation of TLR7 induces high levels of type I IFNs.
HPV: from infection to cancer.
Particle size and activation threshold: a new dimension of danger signaling.
Imiquimod (Aldara) is commonly used topically to treat warts on the skin of the genital and anal areas. However, Imiquimod does not cure warts, and new warts may appear during treatment. Imiquimod does not fight the viruses that cause warts directly, however, it does help to relieve and control wart production. Imiquimod is a topical immune response modifier that acts as a TLR7 agonist. Study shows that combination of locally delivered imiquimod with systemic anti-CD40 immunotherapy drive systemic antitumor immune responses.
The antiviral activity of Toll-like receptor 7 and 7/8 agonists.
Mechanism of action and other potential roles of an immune response modifier.
The immune system counters with too little of a Th1 response, the virus remains concealed and the infection becomes active or chronic. It is well established that immune functioning decreases as we age. During aging, a Th2 dominant immune response becomes established. A Th1 immune response dominates at an earlier age. A Th2 dominant immune response cannot protect us from most infections and cancer.
Dendritic cells (DCs) are potent antigen presenting cells (APCs) that possess the ability to stimulate naive T cells. They comprise a system of leukocytes widely distributed in all tissues, especially in those that provide an environmental interface. DCs are derived from bone marrow progenitors and circulate in the blood as immature precursors prior to migration into peripheral tissues. Within different tissues, DCs differentiate and become active in the taking up and processing of antigens (and their subsequent presentation on the cell surface linked to major histocompatibility (MHC) molecules. Upon appropriate stimulation, DCs undergo further maturation and migrate to secondary lymphoid tissues where they present antigen to T cells and induce an immune response. When naive CD4 T cells encounter dendritic cells, they can develop into the following:
In order to stimulate the production of CD4 T cells that promote cell mediated immunity, dendritic cells must secrete IL-12. If they secrete IL-4, IL-5 and IL-13 instead, the CD4 T cells will promote the secretion of antibody. On the other hand, if the dendritic cells secrete IL-10, tolerance or immune non-responsiveness will occur.
The good news 90% of people with a balanced immune system will clear the virus, without medical treatment, within two years of becoming infected. Smokers or those with suppressed immune systems may need to wait longer. There are many natural agents available to help restore balance in an underactive Th1 immune response. These include:
Omega-3 fatty acids, monounsaturated fats found in olive and hazelnut oils, vitamin A cod liver oil, L-Glutamine, Inosine, Silica, digestive enzymes, friendly intestinal flora or soil based organisms (SBOs), ginseng (Red Korean or concentrated Siberian Ginseng extract), chlorella (spirulina and some other sea vegetables may have similar benefits), thyroid hormones, garlic (raw or aged extract), L-Glutathione (or products that raise levels), DHEA or AED (androstendiol), UV-A light, vitamin E, transfer factor (antigen specific) – protein immunomodulators extracted from colostrum, colostrum, low dose naltrexone, IP6, lentinian and certain other mushrooms, Thymus extracts, licorice root, dong quai, beta 1,3-glucan, noni, neem, gingko biloba, exercise, water (to aid detoxification), a positive attitude and prayer, the ability to forgive and be compassionate, and having long-term goals.
Factors that induce Th2 and suppress Th1 immune reaponse: Processed, heated vegetable oils high in trans-fatty acids and linoleic acid (safflower, soy, canola, corn and sunflower), glucose (white sugar), asbestos, lead, mercury and other heavy metals, pesticides, air and water pollutants, progesterone, prednisone, morphine, tobacco, cortisol (in high doses), HIV, candida albicans, HCV, E coli and many other pathogens, continuous stress, thalidomide, UV-B light, pregnancy, melatonin (conflicting research suggests that high levels induce Th2 while very small amounts induce Th1), alcohol (animals studies show that ethanol definitely suppresses Th1 and induces Th2; beer was not tested and there are some indications it may help), streptococcus thermophilis (sometimes found in yoghurt), 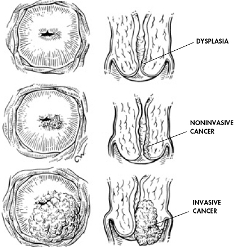 candidiasis, circulating immune complexes (CICs – caused by a combination of leaky gut syndrome and poor digestion of proteins due to a lack or HCl and digestive enzymes), sedentary lifestyle, negative attitudes, low body temperature, acid saliva pH, chronic insomnia, inability to dream, weight lifting, and steroids (for muscle gain).
candidiasis, circulating immune complexes (CICs – caused by a combination of leaky gut syndrome and poor digestion of proteins due to a lack or HCl and digestive enzymes), sedentary lifestyle, negative attitudes, low body temperature, acid saliva pH, chronic insomnia, inability to dream, weight lifting, and steroids (for muscle gain).
Finally, the great news is that it’s quite possible to solve all these problems and cure your chronic HPV infections, genital warts and cervical dysplasia naturally. We have developed a natural Th1 immune modulator/enhancer “Hepazym™”, and succeeded in clinical treatment of primary HPV-16 and HPV-18 infection of the cervix in women. Don’t be depressed by chronic HPV infections. It can be cured. Hepazym™ has demonstrated how new natural treatment way and approaches make it possible to not only cure, but also to prevent HPV infections.